
The low specific activity Bqg can be explained with the large half-life of the isotopes. Therefore, it should not be consumed or handled with bare hands. Uranium is, however, chemically toxic (as are all heavy metals). The fission pathway takes advantage of huge, bloated, radioactive nuclei at the end of the Periodic Table of the Elements, such as uranium or plutonium (elements #92 and #94). 
Is there fission in stars?īy definition, a star is an object that “burns” hydrogen via nuclear fusion. Thus mass is destroyed in the fusion reaction, and energy is released (see Figure 2). … The larger nucleus has a greater binding energy and less mass per nucleon than the two that combined. We know that all nuclei have less mass than the sum of the masses of the protons and neutrons that form them. Normally, fusion is not possible because the strongly repulsive electrostatic forces between the positively charged nuclei prevent them from getting close enough together to collide and for fusion to occur.Īdvertisement Why is mass lost in nuclear fusion? This indirectly results in a greatly increased energy yield, i.e., the bomb’s “power.” This type of weapon is referred to as a hydrogen bomb, or H-bomb, because it employs hydrogen fusion. Is a hydrogen bomb fission or fusion?Ī thermonuclear weapon is a nuclear weapon designed to use the heat generated by a fission bomb to compress a nuclear fusion stage. … Thus, the same naturally occurring elements that can be found on Earth, up to and including elements as heavy as uranium, are also present in the Sun. The process of nuclear fusion combines hydrogen atoms to produce helium and the energy that keeps the Sun shining. The gamma radiation created deep within the sun is absorbed and re-emitted by other atoms as it works its way toward the surface. Gamma radiation in high doses is potentially lethal to life on Earth, but the sun releases relatively little gamma radiation. When neutrons are released during the fission process, they can initiate a chain reaction of continuous fission which sustains itself. In order to initiate most fission reactions, an atom is bombarded by a neutron to produce an unstable isotope, which undergoes fission. Although uranium is about 100 times more common than silver, U-235 is relatively rare. … Nuclear power plants use a certain kind of uranium, referred to as U-235, for fuel because its atoms are easily split apart. Uranium is the fuel most widely used by nuclear plants for nuclear fission. Why only uranium is used in nuclear fission? 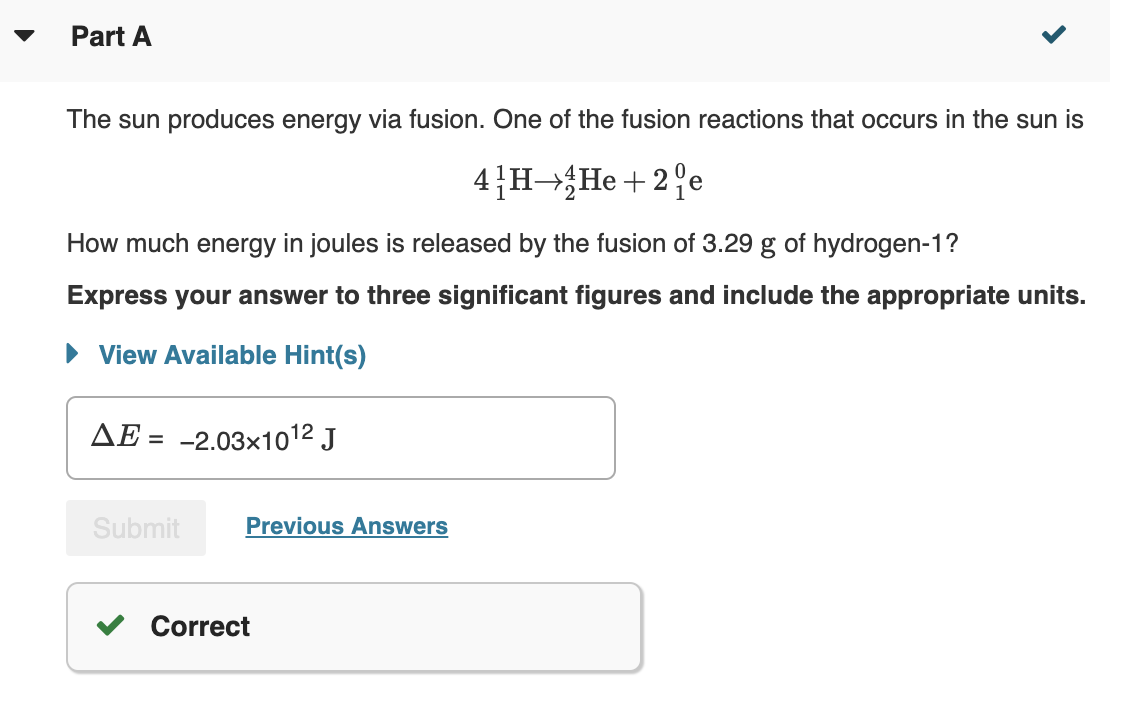
Nuclear fission energy is also use as a power source for propelling submarines and some type of surface vessels. Nuclear fission is used to produce some less common radioisotopes like cesium-137 (Cs-137) by using uranium-235 which is used in photographic sources. Because it is so massive, it has immense gravity, enough gravitational force to hold all of that hydrogen and helium together (and to hold all of the planets in their orbits around the sun). It’s a massive collection of gas, mostly hydrogen and helium. And, in about 5.4 billion years, the Sun will run out of hydrogen fuel, marking the end of its main sequence phase. Some 3.5 billion years from now, the Sun will be 40% brighter than today. The specific type of fusion that occurs inside of the Sun is known as proton-proton fusion. The energy from the Sun – both heat and light energy – originates from a nuclear fusion process that is occurring inside the core of the Sun. In this example, a neutron reacts with uranium-235 to give krypton-92, barium-141, and 3 neutrons. An example would be in nuclear power plants, where uranium is decayed into other substances.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
